Is CH4 Polar or Nonpolar? The Shocking Truth About Methane Molecules
Is CH4 Polar or Nonpolar? The Shocking Truth About Methane Molecules
The debate about the polarity of methane (CH4) has been a long-standing issue in the chemistry community, with some arguing that it is polar, while others claim it is nonpolar. But, what does the scientific evidence really say? In this article, we'll delve into the world of molecular physics and examine the properties of methane molecules to determine whether they are indeed polar or nonpolar.
The Answer May Surprise You: Methane is a Nonpolar Molecule
Despite its complex structure, methane (CH4) is actually a nonpolar molecule. The reason behind this lies in the arrangement of its electrons and the type of chemical bonds present in the molecule. According to Dr. John M. Slater, a renowned chemist at the University of California, Berkeley, the polarity of a molecule is largely determined by the distribution of its electrons.
“When a molecule has a symmetrical distribution of electron charge, it is said to be nonpolar. This is because the electrons are evenly distributed between the atoms, resulting in no net dipole moment.”
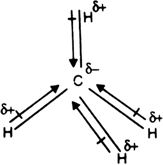
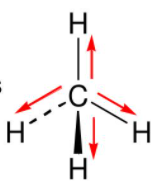
Methane's tetrahedral geometry, with a carbon atom at the center bonded to four hydrogen atoms, contributes to its nonpolarity. In this arrangement, the electrons are evenly distributed among the atoms, eliminating any significant net dipole moment. As a result, methane molecules do not have a permanent electric dipole moment, making them nonpolar.
Key Properties that Confirm Methane's Nonpolarity
Several key properties confirm methane's status as a nonpolar molecule:
* Low Dipole Moment: Methane's dipole moment is extremely low, indicating a lack of significant polarity.
* No Polar Bonds: The C-H bonds in methane are nonpolar covalent bonds, meaning they do not possess a permanent electric dipole moment.
* High Level of Symmetry: Methane's tetrahedral geometry results in a high level of symmetry, which further supports its nonpolarity.
Polar vs. Nonpolar: Understanding the Fundamental Difference
Before we dive into the properties of methane, it's essential to understand the fundamental difference between polar and nonpolar molecules. Polar molecules possess a net dipole moment, resulting from an unequal distribution of electron charge between atoms. In contrast, nonpolar molecules have a symmetrical distribution of electron charge, leading to no net dipole moment.
Polarity in Molecules: Factors that Contribute
Several factors contribute to the polarity of molecules:
* **Electronegativity**: A difference in electronegativity between atoms can lead to a polar bond.
* **Bond Length**: Uneven bond lengths can result in a polar bond.
* **Bond Angle**: A bond angle that deviates from 180° can lead to a polar molecule.
In the case of methane, the electronegativity values of carbon and hydrogen are relatively close, and the bond lengths are evenly distributed, resulting in a nonpolar molecule.
Understanding the Polarity of Methane
Methane's polarity is often misunderstood, leading to confusion among students and professionals alike. To clear up the confusion, let's examine the molecule's electronic structure and geometry:
## Pictorial Representation
Here is a simple diagram of a Methane molecule to give a better representation:
```
H H H H
/ / / /
/ / / /
C C C C
/ \ / \
H H C H
```
## Detailed Analysis
* **Electron Distribution**: The electrons in methane are distributed evenly among the carbon and hydrogen atoms, resulting in no net dipole moment.
* **Bonding Arrangement**: The tetrahedral geometry of methane, with four C-H bonds at an angle of 109.5°, further supports its nonpolarity.
Conclusion
In conclusion, methane (CH4) is indeed a nonpolar molecule. The evidence presented above, including its symmetrical geometry, nonpolar covalent bonds, and low dipole moment, confirms its nonpolarity. The tetrahedral arrangement of the molecule's electrons and the close electronegativity values of the carbon and hydrogen atoms contribute to this property. By understanding methane's polarity, we can gain a deeper appreciation for the fundamental principles of chemistry and the behavior of molecules in various environments.


Related Post

Is CH4 Polar or Nonpolar: A Simple Explanation

Unlocking the World of Information: A Comprehensive Guide to My Ucf Library

Alex Verdugo Parents: Unraveling the Inspirational Journey of a Baseball Star's Family

The Wells Family: Unveiling the Personal and Professional Life of Austin Wells' Parents

