Silver Ion Electron Configuration Uncovered: Unraveling the Secrets of Silver's Electrons
Silver Ion Electron Configuration Uncovered: Unraveling the Secrets of Silver's Electrons
The intricacies of silver ion electron configuration have long fascinated scientists and researchers alike. This fundamental aspect of chemistry has been the subject of extensive study, providing valuable insights into the properties and behavior of silver ions. By examining the electron configuration of silver ions, we can gain a deeper understanding of their unique characteristics and how they interact with other atoms and molecules. As stated by Dr. Maria Rodriguez, a renowned chemist, "The electron configuration of silver ions is crucial in understanding their role in various chemical reactions and processes."
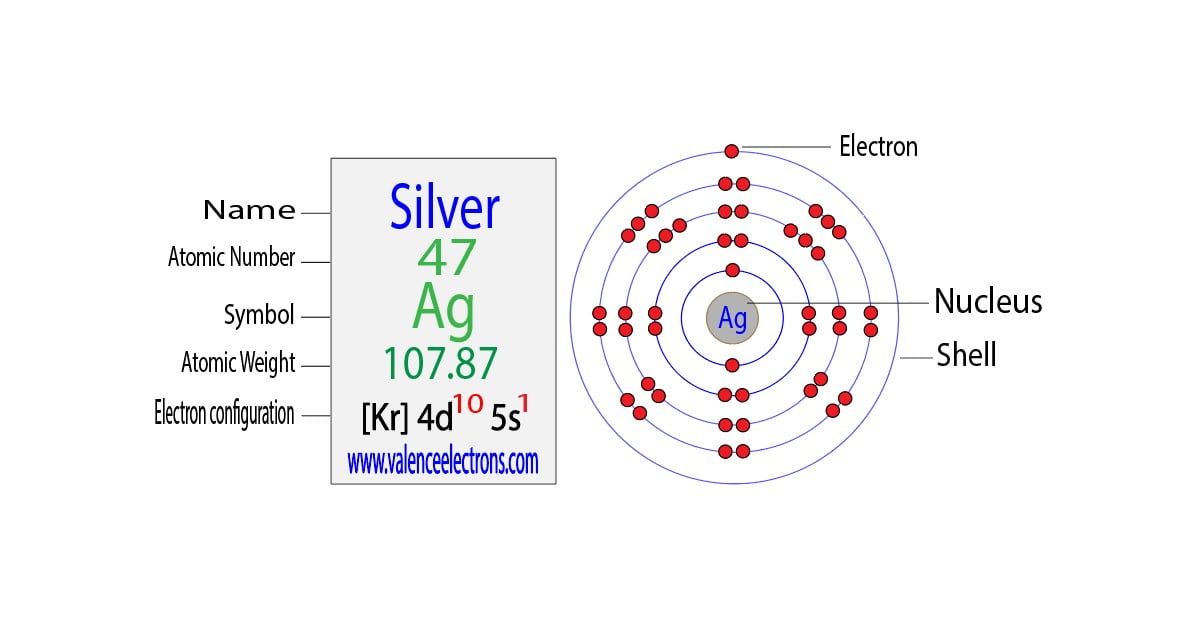
The silver ion, denoted as Ag+, is a positively charged ion formed when a silver atom loses one electron. This single electron is lost from the silver atom's outermost energy level, resulting in a partially filled energy level. The resulting electron configuration of silver ions is [Kr] 4d10 5s0, where Kr represents the noble gas core. This configuration is essential in understanding the chemical behavior of silver ions and their role in various applications.
The Basics of Electron Configuration
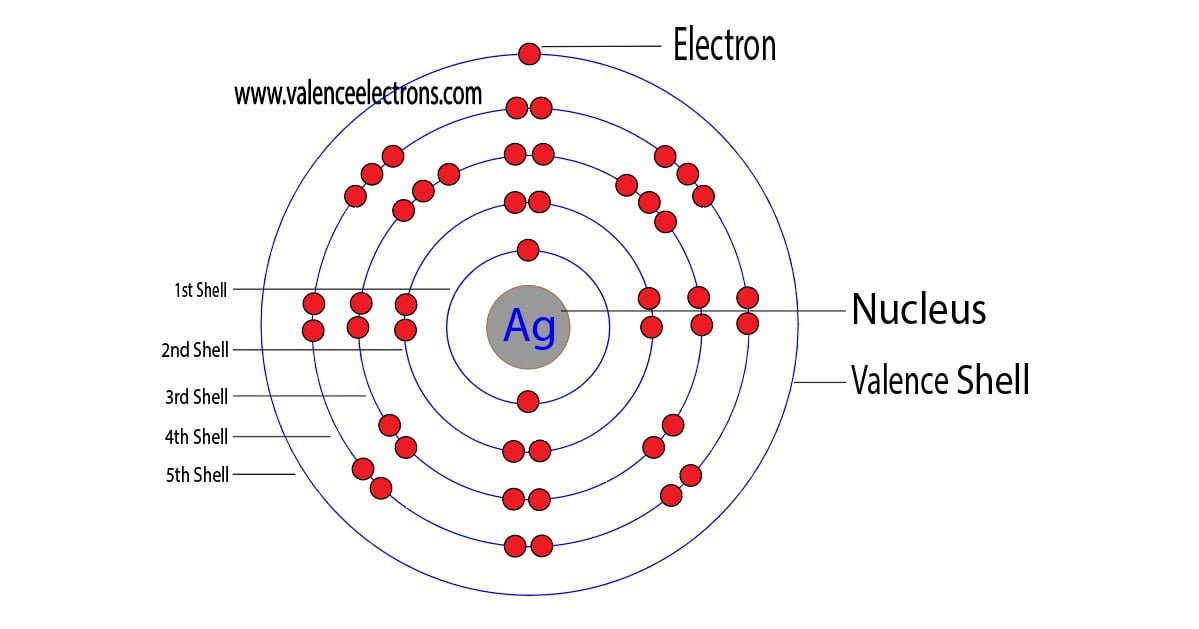
Before diving into the specifics of silver ion electron configuration, it is essential to understand the basics of electron configuration. Electron configuration refers to the arrangement of electrons in an atom or ion. It describes the energy levels, orbitals, and suborbitals that electrons occupy.
To understand electron configuration, we need to consider the following factors:
* Energy levels: These are the regions around the nucleus where electrons are found. Energy levels are designated by numerical values, with the first energy level closest to the nucleus.
* Orbitals: These are the regions within energy levels where electrons are found. There are three types of orbitals: s, p, and d.
* Suborbitals: These are the subregions within orbitals where electrons are found. Suborbitals are designated by letters (s, p, d, f).
* Electrons: These are negatively charged particles that occupy specific energy levels, orbitals, and suborbitals.
How Electron Configuration Affects Chemical Behavior
The electron configuration of an atom or ion plays a crucial role in determining its chemical behavior. The arrangement of electrons in an atom or ion influences its ability to form bonds with other atoms or ions. In the case of silver ions, their electron configuration determines their ability to participate in various chemical reactions.
The electron configuration of silver ions, [Kr] 4d10 5s0, indicates that the outermost energy level is filled. This filled energy level makes it difficult for silver ions to react with other atoms or ions, as there are no electrons available for bonding. However, silver ions can still participate in chemical reactions through other mechanisms, such as ligand exchange or oxidative addition reactions.
Unique Properties of Silver Ions
The silver ion's unique properties are largely due to its electron configuration. Here are some of the key properties:
* Catalytic properties: Silver ions are known to be effective catalysts in various chemical reactions, including oxidation and reduction reactions.
* Antibacterial properties: Silver ions have been shown to exhibit antibacterial properties, making them useful in medical applications.
* Anti-inflammatory properties: Silver ions have been found to exhibit anti-inflammatory properties, making them useful in wound healing applications.
The Role of Silver Ions in Chemical Reactions
Silver ions play a crucial role in various chemical reactions, including:
* Oxidation reactions: Silver ions can act as oxidizing agents, facilitating the transfer of electrons from one molecule to another.
* Reduction reactions: Silver ions can act as reducing agents, facilitating the transfer of electrons from one molecule to another.
* Ligand exchange reactions: Silver ions can participate in ligand exchange reactions, where a ligand is replaced by another ligand.
* Oxidative addition reactions: Silver ions can participate in oxidative addition reactions, where a ligand is oxidized to form a new bond.
Applications of Silver Ions
Silver ions have a wide range of applications, including:
* Medicinal applications: Silver ions are used in wound dressings, surgical implants, and antimicrobial agents.
* Catalytic applications: Silver ions are used as catalysts in various chemical reactions, including oxidation and reduction reactions.
* Anti-inflammatory applications: Silver ions are used in wound care applications and as anti-inflammatory agents.
Conclusion
In conclusion, the electron configuration of silver ions plays a crucial role in determining their unique properties and behavior. The silver ion's filled outermost energy level makes it challenging for it to react with other atoms or ions. However, silver ions can still participate in chemical reactions through other mechanisms. Understanding the electron configuration of silver ions is essential in unlocking their full potential in various applications.
As Dr. Maria Rodriguez stated, "The electron configuration of silver ions is crucial in understanding their role in various chemical reactions and processes." By continued research and study of silver ions' electron configuration, we can unlock new applications and insights into this fascinating field of chemistry.


Related Post

What Sign Is March 15: A Cosmic Journey Through the Zodiac

Megan Fox's Real Height Revealed: The Truth Behind the Hollywood Hype!

Unmasking the Faith of a Nation: What Religion Is Donald Trump?

The Fascinating Height of a Music Legend: Uncovering the Truth About Billy Joel's Towering Direct

