Sn1 Reactions The Ultimate Guide With Real Life Examples
Sn1 Reactions The Ultimate Guide With Real Life Examples
Sn1 reactions, short for substitution, unimolecular reactions, are a type of organic reaction where a molecule gains a nucleophile to form a new bond, while releasing a leaving group from the original molecule. This process is fundamental in various fields, including organic chemistry, pharmaceuticals, and materials science. In this article, we will delve into the world of Sn1 reactions, exploring their mechanisms, types, real-life examples, and applications.
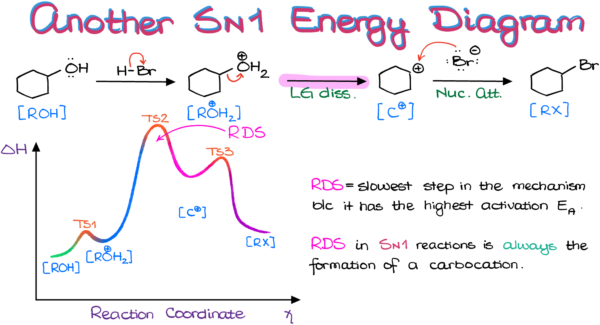
Sn1 reactions are characterized by their unimolecular intermediate, which is formed when a leaving group departs from the substrate. The general equation for an Sn1 reaction is:
where Rx is the reactant, Y is a leaving group, and X- is the set of electron pairs.
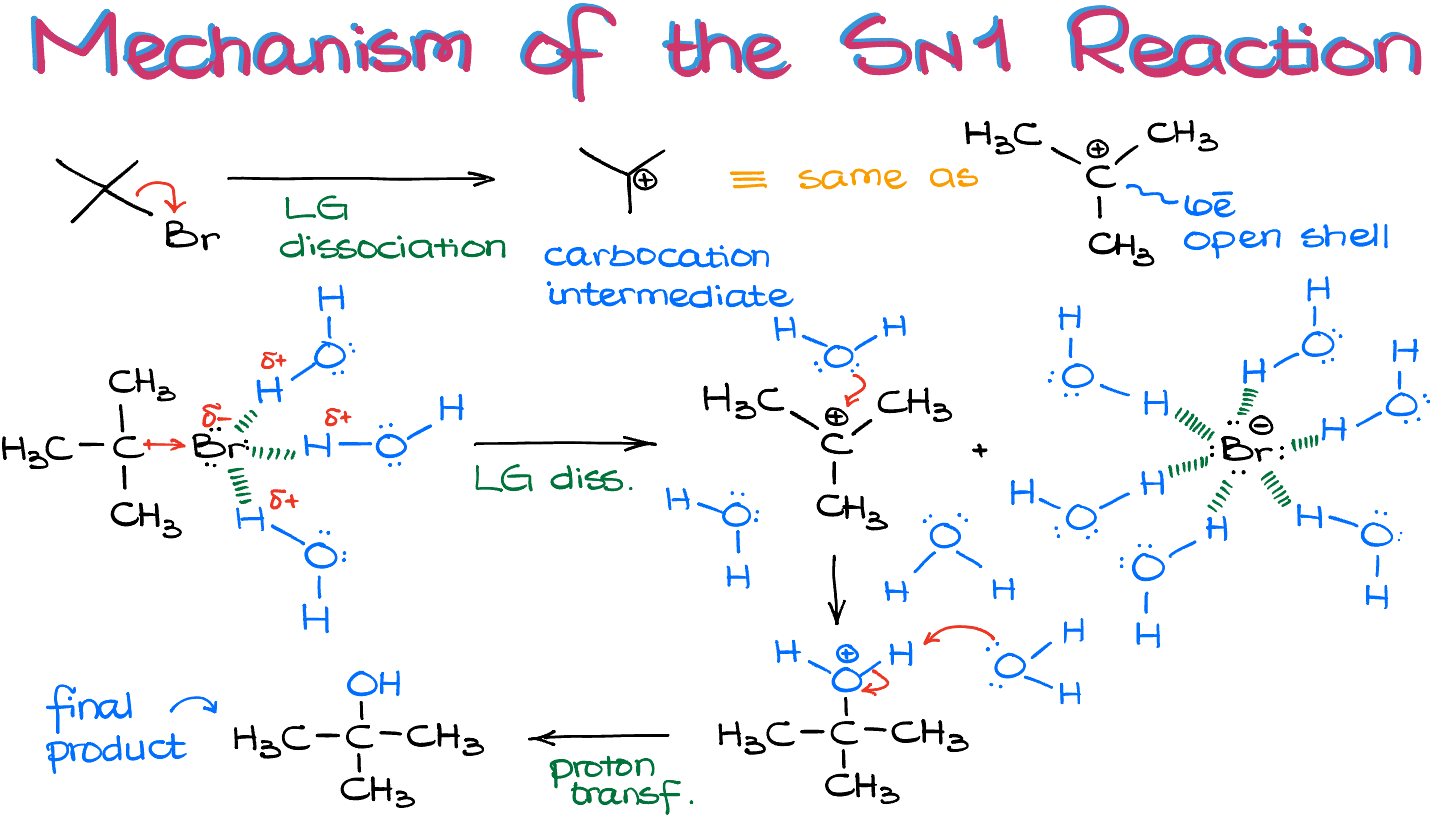
The first step in an Sn1 reaction is the departure of the leaving group, which creates a carbocation. The carbocation is a highly reactive intermediate, and it quickly reacts with a nucleophile (Y-), typically a weak basic species, to form the new bond. The overall process is unimolecular, meaning it occurs in one step, which is a key characteristic of Sn1 reactions.
Types of Sn1 Reactions
There are several types of Sn1 reactions, each with its unique characteristics and applications. Some of the most common types include:
*
Aliphatic Sn1 Reactions
Aliphatic Sn1 reactions involve the substitution of an aliphatic radical with an electrophile. This type of reaction is common in the synthesis of alkanes, alkenes, and alkynes. An example is the reaction of ethyl bromide with iodide,
*
Aromatic Sn1 Reactions
Aromatic Sn1 reactions involve the substitution of an aromatic radical with an electrophile. This type of reaction is common in the synthesis of aromatics, particularly in the production of pharmaceuticals. An example is the reaction of benzene with bromine,
*
Sn1 Reactions in the Presence of a Catalyst
Sn1 reactions can occur in the presence of a catalyst, which accelerates the reaction rate without being consumed. Common catalysts include acid (HCl, H2SO4, etc.) and base (KOH, NaOH, etc.). For example, the reaction of an allylic chloride with hydroxide ion in the presence of sodium iodide,
Real-Life Examples
Sn1 reactions have numerous applications in various fields, including pharmaceuticals, materials science, and biotechnology. Here are a few examples:
*
Pharmaceuticals
Sn1 reactions play a crucial role in the synthesis of many important pharmaceuticals. For example, the conversion of a methylsulfone to a chloro derivative using chloramine-T,
*
Materials Science
Sn1 reactions are essential in the development of new materials, such as polymers and nanomaterials. For instance, the cationic polymerization of isobutylene to polyisobutylene by using Sn1 reaction of isobutylene with a Lewis acid
These examples demonstrate the versatility of Sn1 reactions in various fields. By controlling the reaction conditions, such as temperature, pressure, and concentration, chemists can fine-tune the outcome of Sn1 reactions to produce specific products with tailored properties.
Applications and Limitations
Sn1 reactions have numerous applications in various industries. However, they also have some limitations, including:
*
Regioselectivity
Sn1 reactions can be regioselective, meaning they can produce different products with varying ratios, depending on the reaction conditions and the substrate.
*
Stereochemistry
Sn1 reactions can also be stereoselective, meaning they can produce specific stereoisomers. However, the reaction can lead to mixtures of products due to the reversed configuration of new bonds.
*
Side Reactions
Sn1 reactions can be prone to side reactions, such as elimination reactions, competing with substitution, leading to potential loss of valuable products.
Conclusion
Sn1 reactions are an essential area of study in organic chemistry, encompassing various mechanisms and types of reactions. By understanding the principles and limitations of Sn1 reactions, researchers and chemists can develop new methods for the synthesis of complex molecules, leading to a better understanding of chemical systems. This knowledge can also be leveraged for the development of new materials and pharmaceuticals. With their diverse applications and rich history, Sn1 reactions continue to play a significant role in modern chemistry research.


Related Post

Unlock the Best of Hy Vee Deals Of The Day: Insider Secrets to Saving Big!

Espn Player Rankings Fantasy Football: The Ultimate Guide to Dominating Your League

Uncovering the Hidden Gems of Rockhound Gulch

Unmasked: The Turbulent 50-Year-Old Career of Eminem, the Real Slim Shady

