The Hidden Gem of Chemistry: Lewis Structure For So4 2
The Hidden Gem of Chemistry: Lewis Structure For So4 2
The Lewis structure for SO4 2, also known as sulfuric acid, is a fundamental concept in chemistry that has far-reaching implications for various fields, from organic synthesis to environmental science. This diatomic molecule is often overlooked but plays a crucial role in many chemical reactions and processes. By understanding the Lewis structure of SO4 2, researchers and chemists can better comprehend its properties, reactivity, and potential applications. In this in-depth exploration, we'll delve into the intricacies of the Lewis structure for SO4 2 and its significance in chemistry.
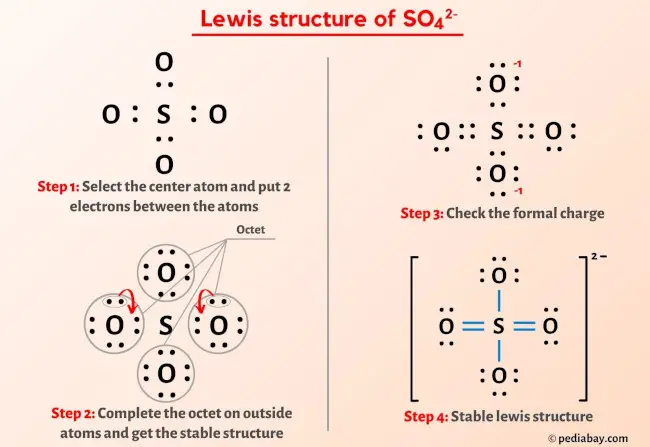
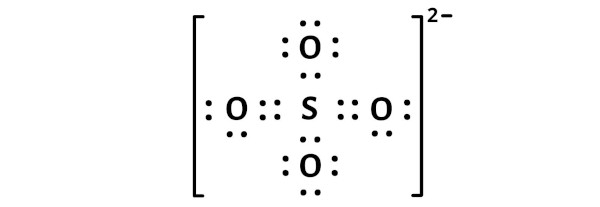
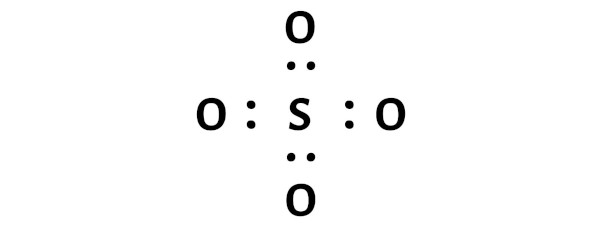
The Lewis structure of a molecule is a two-dimensional representation of the distribution of electrons within a molecule. It provides a visual representation of the arrangement of electrons in a molecule, which is essential for understanding its chemical properties and reactivity. The Lewis structure for SO4 2 consists of one sulfur atom bonded to four oxygen atoms, with two additional oxygen atoms bonded to the sulfur atom through single covalent bonds. This unique arrangement of electrons and bonds gives SO4 2 its distinctive chemical properties.
The structure of SO4 2 can be visualized using a Lewis dot diagram, which consists of a sulfur atom surrounded by four oxygen atoms, each with two pairs of electrons and one pair of lone pairs. The sulfur atom also has two pairs of electron pairs, resulting in a total of eight electrons in its outermost energy level. The Lewis structure for SO4 2 is often represented as:
S=O-O-O-O
This structure reveals that SO4 2 has a trigonal pyramidal arrangement of electron pairs around the sulfur atom. The symmetry and distribution of electron pairs make it an essential concept in understanding the reactivity of SO4 2.
Moreover, the Lewis structure of SO4 2 has garnered attention in the field of organic chemistry due to its application in sulfur-based reagents. In the field of organic synthesis, sulfur-based reagents are widely used as catalysts or intermediates for various reactions. Understanding the Lewis structure of SO4 2 can provide valuable insights into the mechanisms of these reactions and improve the efficiency of the process.
The Lewis structure for SO4 2 can also be used to predict its acidity. In chemistry, acidic molecules are defined as species that donate a proton (H+), making them a crucial component in acid-base reactions. By examining the Lewis structure for SO4 2, researchers can determine its acidity constants (pKa) and use this information to predict its behavior in various chemical reactions. As a result, the Lewis structure for SO4 2 plays a pivotal role in understanding acid-base chemistry and its applications in various fields.
Historical Background and Importance
The concept of the Lewis structure for SO4 2 has been discussed in various scientific communities since its development by Gilbert N. Lewis in 1916. Lewis, an American chemist, is best known for his work on the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve an octet configuration. The Lewis structure for SO4 2 was one of the earliest examples used to demonstrate the application of the octet rule in predicting molecular geometry and reactivity.
The Lewis structure for SO4 2 has been extensively studied in various fields, including chemistry, physics, and biology. Its significance lies in its ability to explain the chemical properties and reactivity of the molecule, which is crucial for understanding various chemical reactions and processes.
The SO4 2 molecule is a fundamental example of a Lewis acid-base reaction. According to the Lewis definition, a Lewis acid is an electron pair acceptor, while a Lewis base is an electron pair donor. The Lewis structure for SO4 2 shows the sulfur atom as an electron pair acceptor, which makes it a Lewis acid. This characteristic makes SO4 2 a key component in many acid-base reactions, influencing the reaction mechanisms and products.
In addition to its chemical properties, the Lewis structure for SO4 2 has been studied in the context of biological systems. In recent years, the study of sulfur-based systems has gained attention due to the increasing interest in sulfur-based reagents and the potential applications in medicine and other fields.
The Lewis structure for SO4 2 has also drawn attention in the field of materials science. Researchers have explored the use of SO4 2 as a precursor to synthesize novel materials with unique properties. This includes the creation of organic-inorganic hybrid nanomaterials, which have improved mechanical, electrical, and thermal properties.
In conclusion, the Lewis structure for SO4 2 is a fundamental concept in chemistry that has significant implications for various fields. It provides valuable insights into the chemical properties and reactivity of the molecule, making it essential for understanding acid-base reactions and applications in organic synthesis. The historical background and importance of the Lewis structure for SO4 2 demonstrate its significance in chemistry and its far-reaching influence on various scientific disciplines.
The study of the Lewis structure for SO4 2 is an ongoing area of research, with researchers exploring its applications and significance in various fields. Further studies on the properties and reactivity of SO4 2 will undoubtedly shed more light on its potential applications in medicine, materials science, and other areas of chemistry.

Related Post

Harris Teeter Weekly Ad: Unlocking the Best Deals on All Your Faves

Tears Across Borders: Uncovering The Heartbreaking International Condolences Database Behind The Tehran Bombing Of 2026

Invest 94L: The Next Big Storm Brewing in the Atlantic?

The Untold Rich Eisen Net Worth That Shocked The World

