Unraveling the Mystery of the Sulfate Ion: A Deep Dive into the So42- Lewis Structure
Unraveling the Mystery of the Sulfate Ion: A Deep Dive into the So42- Lewis Structure
The world of chemistry is a vast and wondrous place, filled with intricate structures and complex reactions. At the heart of it all lies the humble sulfate ion, a seemingly simple entity with a Lewis structure that has captivated scientists and students alike. But what secrets lie hidden within the So42- Lewis structure, and how can we unlock its mysteries? In this article, we'll take a journey into the world of inorganic chemistry, exploring the history, properties, and applications of the sulfate ion, and delving into the intricacies of its iconic Lewis structure.
The sulfate ion, or So42-, is a polyatomic ion consisting of one sulfur atom bonded to four oxygen atoms. Its Lewis structure, a diagrammatic representation of the ion's electron configuration, reveals a complex web of electrons and bonds that underpin its properties and behavior. But what does this structure really tell us, and how can we use it to better understand the sulfate ion? To answer these questions, we need to take a step back and explore the history and context of the sulfate ion.
The History of the Sulfate Ion
The sulfate ion has been a staple of chemistry for centuries, with early studies dating back to the 18th century. One of the earliest recorded mentions of the sulfate ion can be found in the works of French chemist Antoine Lavoisier, who in 1777 isolated the ion from the mineral gypsum. Since then, the sulfate ion has played a central role in the development of inorganic chemistry, with applications in fields ranging from medicine to materials science.
Properties of the Sulfate Ion
So what makes the sulfate ion so unique? For starters, its Lewis structure reveals a symmetrical arrangement of oxygen atoms around the central sulfur atom, a trait known as tetrahedral symmetry. This symmetry is key to the ion's stability and reactivity, allowing it to form strong bonds with other ions and molecules. But the sulfate ion's properties don't stop there.
* **Charge:** The sulfate ion carries a -2 charge, making it a powerful anion that can form strong ionic bonds with cations.
* **Size:** The sulfate ion has a relatively small size, measuring around 1.4 nm in diameter.
* **Polymorphism:** The sulfate ion can exist in several different forms, including the alpha, beta, and gamma forms.
The Lewis Structure of the Sulfate Ion
Now that we've explored the history and properties of the sulfate ion, it's time to dive into the intricacies of its Lewis structure. This diagrammatic representation of the ion's electron configuration reveals a complex web of electrons and bonds that underpin its behavior.
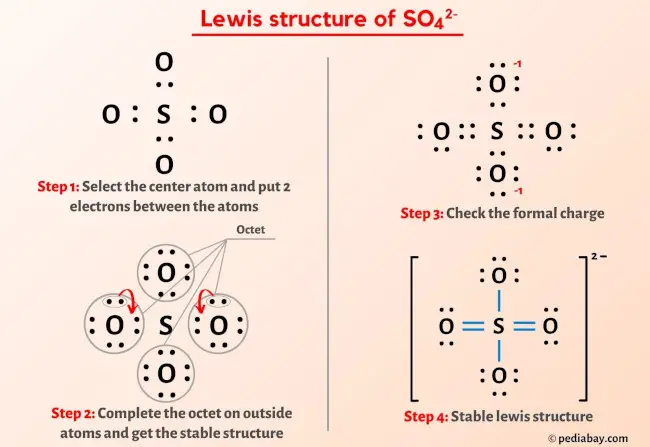
The Lewis structure of the sulfate ion consists of a central sulfur atom bonded to four oxygen atoms, with two single bonds and two double bonds. The sulfur atom is bonded to the four oxygen atoms through a series of covalent bonds, with the oxygen atoms themselves bonded to the sulfur atom through a combination of sigma and pi bonds.
The Significance of the Sulfate Ion's Lewis Structure
So why is the Lewis structure of the sulfate ion so important? For one, it reveals the ion's electronic configuration, providing insights into its reactivity and behavior. By studying the Lewis structure, scientists can better understand the ion's interactions with other ions and molecules, allowing them to develop new materials and compounds with tailored properties.
* **Understanding Bonding:** The Lewis structure of the sulfate ion reveals a complex web of electrons and bonds that underpin its properties and behavior. By studying this structure, scientists can gain a deeper understanding of bonding and reactivity.
* **Predicting Reactivity:** The Lewis structure of the sulfate ion can be used to predict its reactivity with other ions and molecules, allowing scientists to develop new materials and compounds with tailored properties.
* **Designing New Compounds:** By studying the Lewis structure of the sulfate ion, scientists can design new compounds with tailored properties, such as improved strength, conductivity, or optical properties.
Applications of the Sulfate Ion
The sulfate ion has a wide range of applications in fields ranging from medicine to materials science. From the production of fertilizers and detergents to the development of new materials and compounds, the sulfate ion plays a central role in many industries.
* **Medicine:** The sulfate ion is used in the production of many pharmaceuticals, including antibiotics and painkillers.
* **Materials Science:** The sulfate ion is used to develop new materials with tailored properties, such as improved strength, conductivity, or optical properties.
* **Fertilizers:** The sulfate ion is used in the production of fertilizers, providing essential nutrients for plant growth.
Conclusion
In conclusion, the sulfate ion is a complex and fascinating entity with a rich history and a wide range of applications. Its Lewis structure reveals a complex web of electrons and bonds that underpin its properties and behavior, providing insights into its reactivity and behavior. By studying the Lewis structure of the sulfate ion, scientists can gain a deeper understanding of bonding and reactivity, predict its behavior with other ions and molecules, and design new compounds with tailored properties. Whether in medicine, materials science, or fertilizer production, the sulfate ion plays a central role in many industries, making it an essential component of our daily lives.



Related Post

Watch Yankees Game Tonight: Find Your Channel On Optimum and Dive into the World of Baseball

Centre Daily Times Obituaries Past 3 Days: A Glimpse into the Lives of Those Who Left a Lasting Impact

The Rise and Evolution of Los Chapitos Cartel: Understanding the Most Powerful Cartel in Latin America

Diddy Sign: The Rise of a Legendary Entertainment Mogul

