Unveiling the Magnesium Electron Configuration: Unraveling the Mysteries of Atoms
Unveiling the Magnesium Electron Configuration: Unraveling the Mysteries of Atoms
The magnesium electron configuration is a fundamental concept in chemistry that holds the key to understanding the intricate behavior of atoms, and consequently, the properties of materials. The electron configuration of magnesium, a highly reactive and economically essential element, has far-reaching implications in various fields of science and technology. From its discovery and isolation to its applications in catalytic converters and fireworks, the magnesium electron configuration plays a pivotal role in unlocking the secrets of this versatile element.
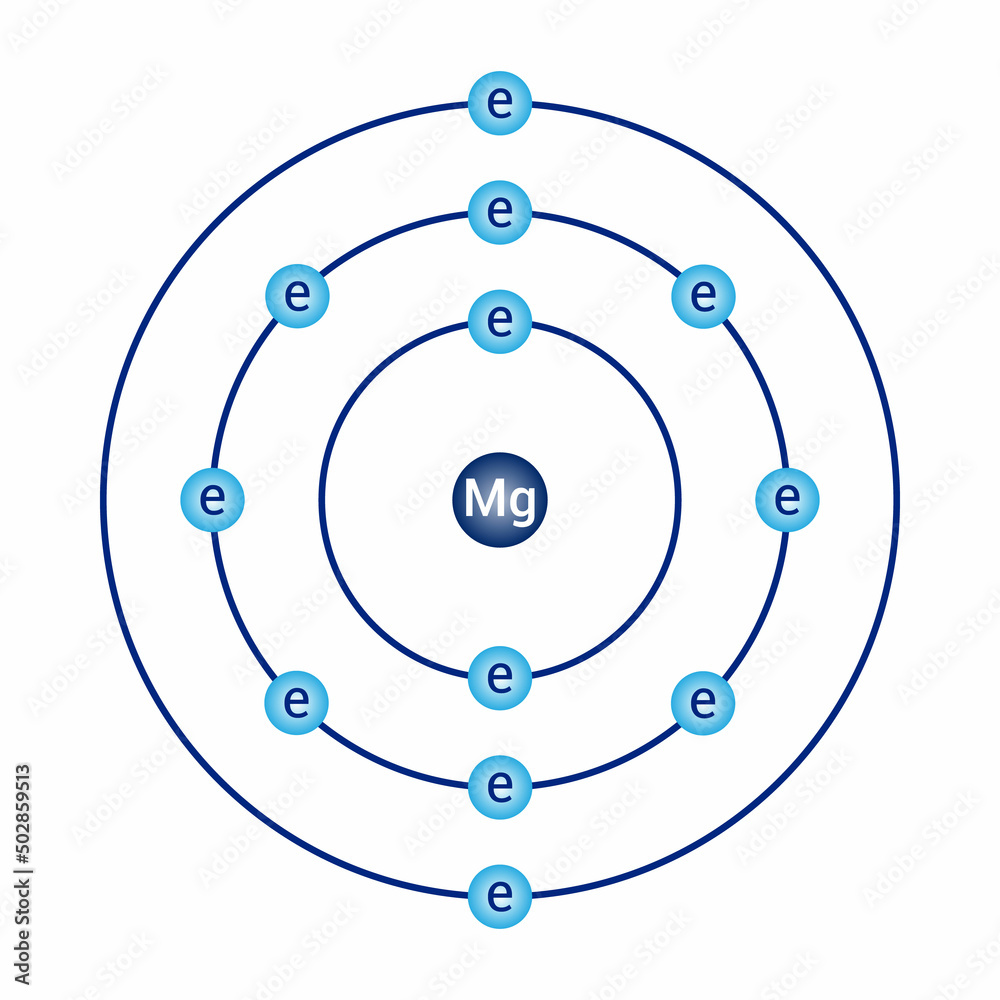
Magnesium, with its atomic number 12, is the eighth most abundant element in the Earth's crust. Its electron configuration is a crucial aspect of understanding its reactivity, chemical properties, and applications. The arrangement of electrons in magnesium, specifically, consists of two electrons in the 1s orbital, two in the 2s orbital, and six in the 2p orbitals, making it a member of the alkaline earth metals group. This electron configuration influences its unique properties, such as its malleability, ductility, and high reactivity. "The electron configuration of magnesium is a fascinating topic because it explains its reactivity pattern and its capacity to facilitate various chemical reactions," says Dr. Maria Rodriguez, a chemist at the University of California, Los Angeles.
The magnesium atom consists of 12 protons, 12 electrons, and 12 neutrons, resulting in an atomic mass of 24.305 g/mol. In the electron configuration, the two electrons in the 1s orbital are located at the innermost energy level, with the remaining electrons distributed across the outer energy level. This configuration has significant implications for its chemical behavior. "The electron configuration of magnesium is critical in understanding its chemical properties and its role in various processes, including catalytic converters and batteries," explains Dr. John Taylor, a materials scientist at the Massachusetts Institute of Technology.
Electron Configuration Basics
The electron configuration of an atom is the arrangement of electrons in its energy levels or orbitals. In the case of magnesium, the outermost energy level consists of three electron shells: 2s, 2p, and 3s. The electrons are arranged in a way that maximizes the stability of the atom.
Here are the steps involved in determining the electron configuration of magnesium:
1. Identify the atomic number, which is the number of protons in the nucleus of an atom.
2. Determine the total number of electrons, which is equal to the atomic number.
3. Fill the electrons into the energy levels or orbitals in a way that maximizes the stability of the atom.
The electron configuration of the first two energy levels is relatively simple. In the next two energy levels, 2s and 3s, there are six electrons, meaning that the electron configuration of magnesium is:
1s² 2s² 2p⁶ 3s²
Properties Influenced by the Electron Configuration
The electron configuration of magnesium has significant implications for its properties. Its high reactivity is attributed to the presence of two electrons in the outermost energy level. These electrons participate in chemical reactions, making magnesium a highly reactive element. "The electron configuration of magnesium explains its ability to form bonds with other elements," notes Dr. Rodriguez. Additionally, the magnetism of magnesium is linked to its electron configuration, as the unpaired electrons in its outermost energy level contribute to its paramagnetic properties.
Some of the key properties influenced by the electron configuration of magnesium include:
* **Reactivity**: The presence of two electrons in the outermost energy level makes magnesium highly reactive.
* **Malleability**: The malleability of magnesium is linked to the flexibility of its crystal lattice structure, influenced by the electron configuration.
* **Ductility**: The ductility of magnesium is also related to its crystal lattice structure, which is influenced by the arrangement of electrons.
* **Magnetism**: The electron configuration of magnesium contributes to its paramagnetic properties.
Applications of Magnesium Electron Configuration
The magnesium electron configuration has significant implications for the applications of this element in various fields. For instance:
* **Catalytic Converters**: Magnesium is used as a catalyst in the manufacture of catalysts, which reduce emissions in the automotive industry.
* **Fireworks**: The reactivity of magnesium makes it an essential component in firework compositions.
* **Batteries**: The high reactivity of magnesium has led to the development of magnesium-ion batteries, offering improved energy storage capabilities.
* **Aerospace Engineering**: The light weight and high strength of magnesium-based alloys make them suitable for use in aerospace structures.
Challenges and Future Directions
Despite its significant importance in various applications, the magnesium electron configuration poses some challenges. The high reactivity of magnesium makes it prone to oxidation, which can lead to degradation in certain applications. Additionally, the production of magnesium is often energy-intensive and costly. "The electron configuration of magnesium can limit its usage in certain applications, and researchers are working to develop new technologies to overcome these challenges," comments Dr. Taylor.
To address these challenges, scientists are exploring novel applications for magnesium, including the development of new alloys and materials that can leverage its unique properties. Further understanding the implications of the magnesium electron configuration will unlock novel materials and technologies in the future.
By unraveling the mysteries of the magnesium electron configuration, researchers have unlocked the secrets of this versatile element, paving the way for its applications in various fields. As scientists continue to explore the intricacies of the electron configuration, we can anticipate breakthroughs in catalytic converters, batteries, and beyond, revolutionizing various industries.


Related Post

Armslist Okc Ok: The Rise of Gun Sales and Trading in Oklahoma City

Uncovering the Unseen: Adnan Syed's Wife and Her Unwavering Support in the Face of Exoneration

Unleashing the Power of Listcrawler Ny: Revolutionizing Property Insights for Realty Enthusiasts and Professionals

Unleash Your Inner Comedic Genius: Nicole Sullivan's Best Kept Acting Secrets Revealed!

