Ch4 Polar Or Nonpolar: Understanding the Chemistry of Hydrocarbons
Ch4 Polar Or Nonpolar: Understanding the Chemistry of Hydrocarbons
The chemistry of hydrocarbons, specifically the polar or nonpolar nature of carbon compounds, has been a topic of interest for scientists and researchers for decades. Hydrocarbons, the building blocks of all life, come in various forms and exhibit unique properties that can be polar or nonpolar in nature. In this article, we will delve into the world of hydrocarbons and explore what makes them polar or nonpolar, and how this affects their behavior and uses.
Carbon, the foundation of all hydrocarbons, has a unique ability to form long chains and rings, creating a vast array of molecules with diverse properties. Hydrocarbons can be classified into several types, including alkanes, alkenes, alkynes, and aromatics, each with its own set of characteristics. However, the polar or nonpolar nature of a hydrocarbon is determined by the presence or absence of electronegative atoms, such as oxygen, nitrogen, or fluorine, attached to the carbon chain.
The Basics of Polar and Nonpolar Molecules
To understand the polar or nonpolar nature of hydrocarbons, it's essential to grasp the basic principles of molecular polarity. Polarity is a measure of the uneven distribution of electrons within a molecule, resulting in a partial positive charge on one side and a partial negative charge on the other. This phenomenon arises when atoms with different electronegativities share electrons, leading to an unequal sharing of electrons and a resulting dipole moment.
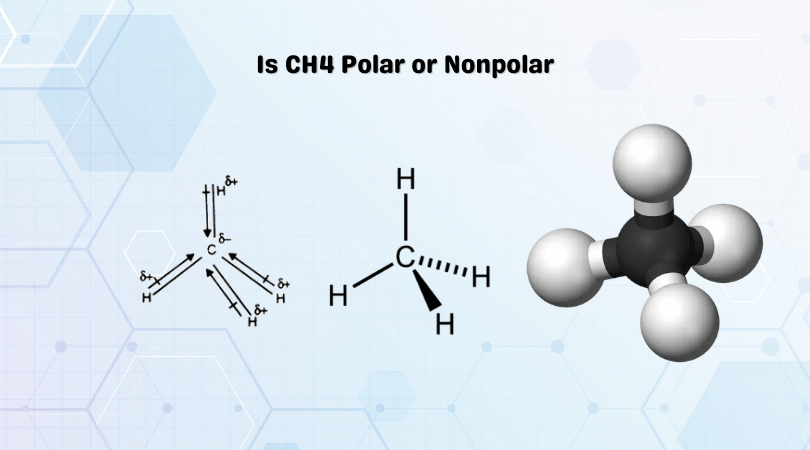
In nonpolar molecules, the electronegativities of the atoms are similar, resulting in an equal sharing of electrons and a symmetrical distribution of charge. Nonpolar molecules tend to be unreactive and have low melting and boiling points. Examples of nonpolar hydrocarbons include methane (CH4), ethane (C2H6), and propane (C3H8).
On the other hand, polar molecules exhibit an unequal sharing of electrons, resulting in a partial positive charge on one side and a partial negative charge on the other. Polar molecules tend to be more reactive and have higher melting and boiling points. Examples of polar hydrocarbons include water (H2O), methanol (CH3OH), and ethanol (C2H5OH).
The Role of Electronegativity in Polarization
Electronegativity, a measure of an atom's ability to attract electrons, plays a crucial role in determining the polar or nonpolar nature of a molecule. Atoms with high electronegativity, such as oxygen, nitrogen, and fluorine, tend to pull electrons closer, resulting in a partial negative charge. Conversely, atoms with low electronegativity, such as carbon and hydrogen, tend to donate electrons, resulting in a partial positive charge.
In hydrocarbons, the presence of electronegative atoms such as oxygen, nitrogen, or fluorine attached to the carbon chain can create a polar molecule. For instance, methanol (CH3OH) is a polar molecule due to the presence of the electronegative oxygen atom. On the other hand, methane (CH4) is a nonpolar molecule due to the absence of electronegative atoms.
Importance of Polar and Nonpolar Hydrocarbons in Industry and Everyday Life
The polar or nonpolar nature of hydrocarbons has significant implications for various industries and aspects of everyday life. Polar hydrocarbons, such as methanol and ethanol, are used as solvents, fuels, and intermediates in the production of pharmaceuticals, dyes, and other chemicals.
Nonpolar hydrocarbons, such as alkanes, are used as fuels, lubricants, and plastics. For instance, gasoline, a mixture of nonpolar hydrocarbons, is used as a fuel for vehicles. Nonpolar hydrocarbons are also used in the production of polymers, such as polyethylene and polypropylene, which are used in packaging, textiles, and other applications.
Environmental and Health Implications
The polar or nonpolar nature of hydrocarbons can have significant environmental and health implications. Polar hydrocarbons, such as methanol and ethanol, can be toxic and have been linked to various health problems, including neurological damage and cancer.
Nonpolar hydrocarbons, such as alkanes, can also have environmental implications. For instance, the release of nonpolar hydrocarbons into the environment can contribute to air and water pollution, harming aquatic life and human health.
Conclusion
In conclusion, the polar or nonpolar nature of hydrocarbons is a complex phenomenon that has significant implications for various industries and aspects of everyday life. Understanding the role of electronegativity in determining molecular polarity is essential for designing new materials and processes. By recognizing the importance of polar and nonpolar hydrocarbons, we can better appreciate the intricate chemistry of hydrocarbons and work towards developing more sustainable and environmentally friendly solutions.
As Dr. Susan Solomon, a leading researcher in the field of atmospheric chemistry, notes, "The polar or nonpolar nature of hydrocarbons is a critical factor in determining their reactivity and fate in the environment. By understanding this phenomenon, we can better develop strategies for mitigating the environmental impacts of hydrocarbon emissions."




Related Post

Is CH4 Polar or Nonpolar: A Simple Explanation
Is CH4 Polar or Nonpolar? The Shocking Truth About Methane Molecules

Kimberly Guilfoyle's Jaw-Dropping Transformation: Before And After

Mo Rocca Married: A Look into the Life of the Prolific Comedian and Commentator

